Why KI?
Part II – The Mammary Glands
In Part I we identified many parts of the body that use iodides (I-). In this section we will look at the mammary glands or breasts. First of 3 parts of the body for which it has been argued requires molecular iodine (I2).
The Breasts Use Iodine
 Iodine concentrating cells includes the mammary glands, placenta, uterus and ovaries.10,11 The breasts are the second major glandular storage site for iodine after the thyroid gland. Women with larger breasts generally require more iodine. Supplementing with iodide is also key for breast cancer prevention and treatment. Iodide has been shown to have anti-tumor effects on mammary tumors.12 The breasts take up iodine the same way that the thyroid does. Iodine concentrating mechanisms of the mammary gland closely resembles those of the thyroid iodide trap and the breast tissue even has the capacity to add iodine to amino acids of cell membranes.13 This makes cells less reactive to free oxygen radicals. For example, peroxidase, a family of enzymes that use iodide to make iodine available for the thyroid gland, is found in the mammary glands.14 Clearly there are mechanisms in place for the breasts to use iodide to create organified iodine for local use.
Iodine concentrating cells includes the mammary glands, placenta, uterus and ovaries.10,11 The breasts are the second major glandular storage site for iodine after the thyroid gland. Women with larger breasts generally require more iodine. Supplementing with iodide is also key for breast cancer prevention and treatment. Iodide has been shown to have anti-tumor effects on mammary tumors.12 The breasts take up iodine the same way that the thyroid does. Iodine concentrating mechanisms of the mammary gland closely resembles those of the thyroid iodide trap and the breast tissue even has the capacity to add iodine to amino acids of cell membranes.13 This makes cells less reactive to free oxygen radicals. For example, peroxidase, a family of enzymes that use iodide to make iodine available for the thyroid gland, is found in the mammary glands.14 Clearly there are mechanisms in place for the breasts to use iodide to create organified iodine for local use.
Human Studies on Breast Tissue: Potassium Iodide Is Better
Russian scientists published results from their study on treating dyshormonal hyperplasia of the mammary glands with KI.15 Dyshormonal hyperplasia means there is a problem with the hormones where abnormal tissue growth is caused by the generation of new cells. They treated 200 human patients with 10-20 mg of iodine with duration ranging from 6 months to 3 years. They observed significant reduction in swelling, pain, hardening, and lumpiness in the breasts. Of the 167 that completed the program, 72% of the patients had shown clinical improvement. No patients experienced any negative side effects. Five patients had ovarian follicular cysts reduced after 5-12 months of supplementing with potassium iodide (KI). Note the amount of time it takes for the body to correct itself when given a maintenance dose of iodine. A shorter time period may not tell the whole story.
Canadian scientists tested various amounts of Lugol’s iodine in three open trials.16 Lugol’s 5% solution was used in 233 human patients for two years in daily amounts ranging from 31-62 mg of iodine. With these higher amounts they were able to achieved clinical improvement in 70% of the patients however 7% of patients experienced negative side effects. Of the 7% of patients that experienced negative side effects, about half experienced iodism. The other half had adverse thyroid functions. The following table summarizes both studies and makes for an easy comparison.
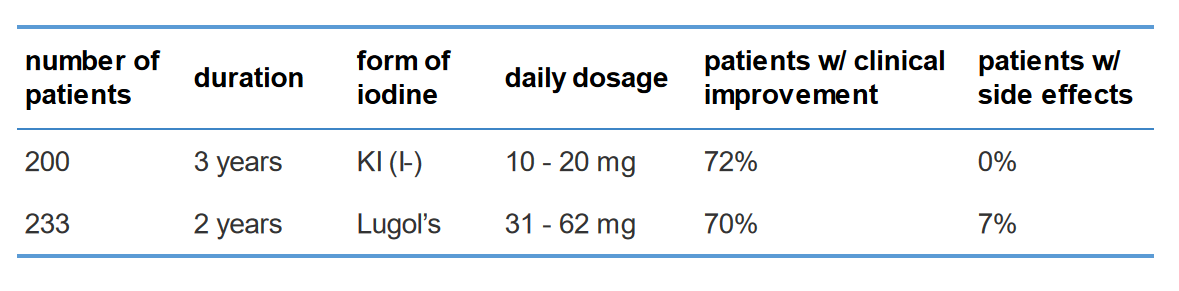
This table makes it clear that potassium iodide (KI) was more effective for breast tissue while also being safer to use. The Lugol’s iodine dosage had to be about three times higher to get an improvement rate that was almost as good as the potassium iodide study. A much lower dose of potassium iodide still proved to be more effective.
Its is also important to note that there were no negative side effects in the potassium iodide study. The Lugol’s study had 7% of patients exhibiting negative side effects which includes adverse thyroid function. Imagine taking an iodine supplement with the chance of it actually impairing your thyroid. Since the body gives thyroid preference for iodine, and the thyroid is the primary user of iodine in the body, we should follow suit and make its safety and healthy function a priority. We can do that while also supplying the breasts with iodine by supplementing with potassium iodide. Potassium iodide (KI) is safe to use.
Potassium iodide is more effective for breast tissue in human patients and has also shown to be effective in reducing ovarian cysts. It will not impair the thyroid and with the recommended maintenance amounts there is virtually no risk of iodism.
Rats Are Less Relevant
We know that human studies will always be more relevant than animal studies. Of the different types of animals used in iodine studies, rats may be the least relevant for our purposes. It has been shown that iodide concentrating mechanisms are present in the mammary glands of lactating guinea-pigs and mice. However rats are exceptional because they do not have the same non-thyroid iodide trapping mechanisms.17 They are different from humans and other animals when it comes to iodide trapping mechanisms in the mammary glands.
Rat Study
We have already shown that the mammary glands concentrate iodide and that abnormal breast tissue is effectively treated with potassium iodide. Let’s look at the main argument given for why the breasts need Lugol’s form.
A short study was done on rats that were fed molecular iodine (I2) and iodide (I-). It suggests that some breast tissues of some of the rats showed more improvement from molecular iodine (I2).18 Here is how the study was set up.

The iodine deficient diet, perchlorate water and injected estrogen were used to create conditions for abnormal breast tissue to form.
Aside from the study being based on rats, here are some concerns and why we think it is not a good basis for drawing conclusions on iodine treatment for human breast tissue.
Different Number of Rats
The first inconsistency you may notice is the number of rats given molecular iodine (I2) were twice as those given potassium iodide (KI). Also, both of these amounts differ from the control group. It is not clear why this was the case.
Estimates and Inconsistent Dosages
The iodine available in the solutions given were said to be ‘similar’. The iodine available in the molecular iodine (I2) solution was estimated. Though it was an estimate, the amount of iodine in the molecular iodine (I2) solution was higher that the iodine available in the potassium iodide (KI) solution. In other words the rats receiving the potassium iodide solution got a lower dose of iodine. How much lower cannot be made certain.
Short Duration (Only 10 days)
The study observes iodine treatment on mammary tissues over a 10 day period. A longer time horizon, as was used in the human studies, is more appropriate for supplementing with iodine. The extent of iodine deficiency we are dealing with has come about over decades of environmental factors and inadequate diets. Iodine supplementation used to correct nutritional deficiencies and treat diseases should be thought of as an ongoing process by which the body is provided with what it needs to heal itself over time. This is more like the eastern approach to medicine. Quick fixes that aim to heal within a few weeks, even months, could make matters worse. This is the old paradigm and the big pharma way of thinking about medicine. Six months to three years seems to be a more appropriate period of time.
Rats Cannot Convey Symptoms of Iodism
Of course, it is much more difficult to detect iodism in rats because they are unable to report its symptoms.
Breast and Thyroid Connection
The mammary glands can effectively compete with the thyroid gland for peripheral iodine.19 A healthier thyroid seems to be related to healthier breasts. It has been well established that the thyroid, the main site for glandular storage and highest concentration of iodine in the body, uses iodide (I-). Iodine is organified locally in both the thyroid and mammary glands. Logic demands then that, if these sites compete for iodine/iodide, then the mammary glands must use iodide. Moreover, before treating mammary glands it may be more effective to first treat the thyroid.20
Preventing Breast Cancer with Potassium Iodine
One of the ways iodine can help prevent breast cancer involves the antioxidant properties of iodide (I-).21 The process of converting iodide to iodine has antioxidant effects on the area. Breast cancer tissue samples suggest impaired and non-functional sodium-iodide symporters (NIS). We must consider that we see impaired function of NIS expression during the initiation of cancer formation.22 Repairing and enhancing NIS symporter functionality in order to get the benefit of iodide in the breast tissues should be made a priority in breast cancer treatment, especially when using iodine supplementation.
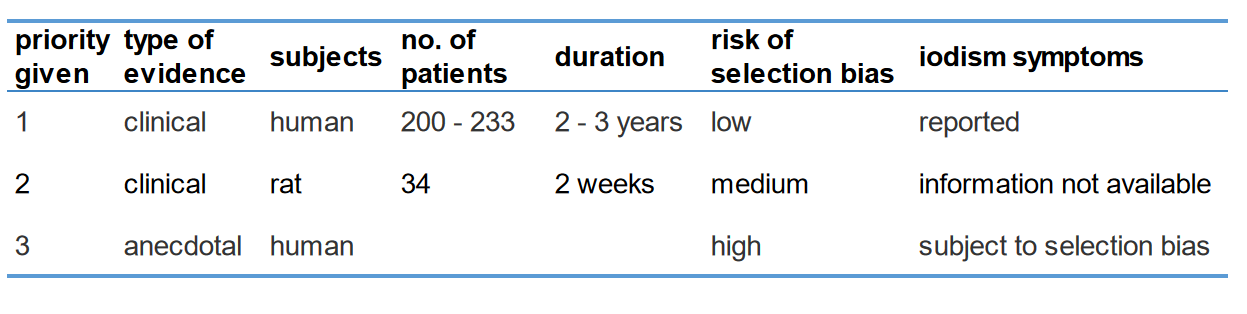
Let us revisit the principles for considering data and evidence.
- clinical evidence is better than anecdotal evidence
- human studies are better than rat studies
- longer studies are better than shorter studies
- more sample or patient data is better than less
- raising a question does not constitute an argument
The first four principles come into play when looking at the studies shown for breast tissue. We took the liberty of assigning a priority value and are confident that you will agree with our assessment. The human clinical studies, having more sample data, a longer duration and lower risk of selection bias, is the clear winner. The evidence it provides should be given priority over the others. That is, potassium iodide (KI) is the preferred form for supplementing when it comes to making iodine available for the mammary glands.
 The evidence shows that the breasts utilize iodide. When using iodine to treat the mammary glands we must consider the state of the thyroid. Is it functioning properly? Is the thyroid getting enough iodine on a regular basis? After the thyroid’s iodine needs are satisfied is there still enough iodine available for the rest of the body? Are the mammary NIS functioning? Are goitrogens being limited? While these variables are addressed, supplementing with potassium iodide will provide greater benefit to the mammary glands and the rest of the body.
The evidence shows that the breasts utilize iodide. When using iodine to treat the mammary glands we must consider the state of the thyroid. Is it functioning properly? Is the thyroid getting enough iodine on a regular basis? After the thyroid’s iodine needs are satisfied is there still enough iodine available for the rest of the body? Are the mammary NIS functioning? Are goitrogens being limited? While these variables are addressed, supplementing with potassium iodide will provide greater benefit to the mammary glands and the rest of the body.
In this section we have shown that potassium iodide (KI) is the preferred form for the mammary glands for the following reasons:
- clinical evidence provided from human studies indicate potassium iodide (KI) is more safe and effective
- clinical evidence from human subjects is more reliable than rat studies
- long-term studies with greater sample data is more reliable than short-term studies with less data
- refuted arguments for molecular iodine (I2) based on rat studies
In Part III we discuss the stomach and the prostate, the two other parts of the body where it has been argued requires I2.
.


